 
Since the year 2011, there have been rapid developments in the field of immunotherapy, and a range of immunocheckpoint inhibitors (ICIs) have been approved for a range of metastatic cancers, including malignant melanoma, lung, kidney, and bladder cancers. In conclusion, intensive studies over an extended range of cancer types and a broadened group of subjects should be carried to solidify the TCR repertoire's position as an immunotherapy biomarker. Recent progress of TCR sequencing technology in tumor research is also discussed. In this article, we reviewed TCR rearrangement mechanisms and analysis methods. The results of some studies are even contrary. 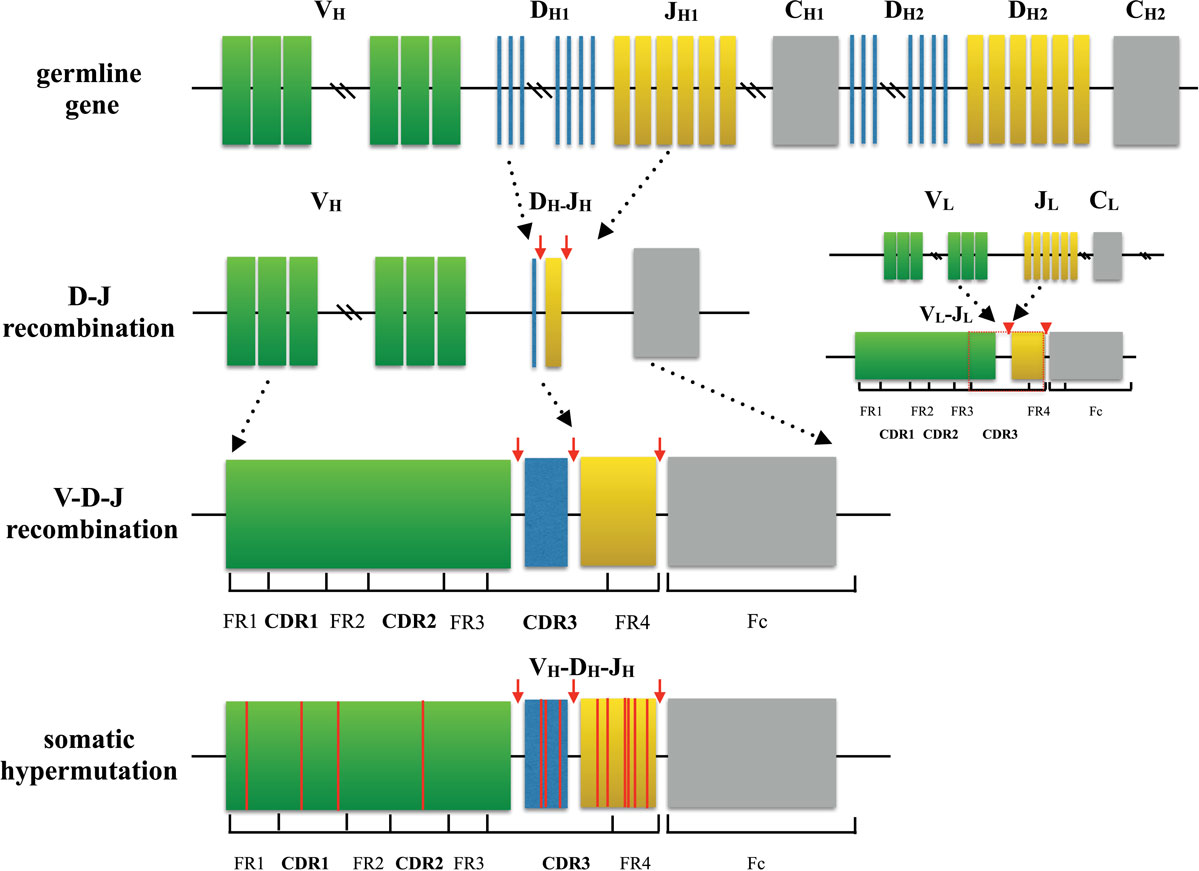 
Still, existing researches are insufficient to clarify the specific clinical implications of TCR dynamic change and the definite role of TCR repertoire diversity during the treatment process. Accurate delineation of the T‐cell repertoire can further the understanding of the immune system response to tumorigenesis. The potential of TCR repertoire as a biomarker for immunotherapy efficacy is also widely studied as TCR repertoire represents different baseline within individuals and shows dynamic change during treatment. Results have shed light on the TCR repertoire differences between cancer patients and healthy control as well as between individual's lesions, paracancer, and peripheral blood samples. Based on this new methodology, discoveries are made across a range of tumor types. Nowadays, next‐generation sequencing technology allows the simultaneous detection of TCR sequences with high throughput, and several evaluation indexes facilitate the measure of TCR repertoire. Thus, TCR repertoire analysis occupied an important position in T‐cell regarding research. The unique rearrangement mechanisms during T‐cell maturation provide great diversity to TCR, ensuring specific recognition between T cells and antigens. Mature T cells can specifically recognize the antigen on the major histocompatibility complex (MHC) molecule through T‐cell receptors (TCR). Preliminary results suggest the barcoded transcripts produced have the potential to recover TCR pairings at scale.T cells, which are involved in adaptive immunity, are essential in the elimination of tumor cells. This method, spatial TCR deconvolution, amplifies TCR transcripts from cells lysed in solution and uses barcoded beads as a molecular beacon to record spatial proximity. Finally, we describe how this framework primed the development of a novel T cell sequencing strategy. By developing a new Bayesian framework, we are able to increase pairing efficiency and computation speed. To do this, we focus on algorithmic approaches to recover TCRα and TCRβ pairings in noisy datasets. In my second aim, we explore integrated experimental and computational approaches for sequencing TCR repertoires. We then use this technique to screen for antigenic peptides derived from multiple pathogen proteomes across eighteen MHC alleles, resulting in a catalog of new antigens to further characterize. This platform utilizes yeast surface display as a means to separate binding peptide from non-binding peptide. In the first aim, we describe a high-throughput tool to identify antigenic peptides capable of being presented on class I MHCs. In this thesis, we discuss two separate advances in immune repertoire sequencing. Our ability to detect disease relies on the heterogeneity found on both sides of this equation, yet technologies to characterize either antigen presentation or TCR diversity at a repertoire scale are rarely economical or scalable. At the center of this lies the interaction between the T cell receptor (TCR) and antigens presented through peptide-major histocompatibility complexes (pMHCs). Our immune system is a complex network of cells and proteins designed to identify and eliminate cancer and infection.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
